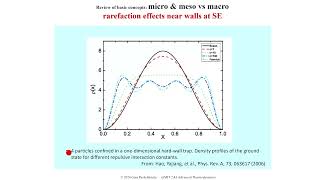
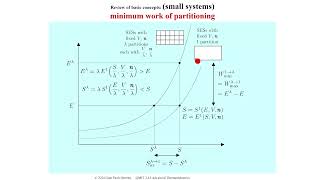
課程簡介
This course is a self-contained concise review of general thermodynamics concepts, multicomponent equilibrium properties, chemical equilibrium, electrochemical potentials, and chemical kinetics, as needed to introduce the methods of nonequilibrium thermodynamics and to provide a unified understanding of phase equilibria, transport, and nonequilibrium phenomena useful for future energy and climate engineering technologies. Applications include second-law efficiencies and methods to allocate primary energy consumptions and CO₂ emissions in cogeneration and hybrid power systems, minimum work of separation, maximum work of mixing, osmotic pressure and membrane equilibria, metastable states, spinodal decomposition, and Onsager’s near-equilibrium reciprocity in thermodiffusive, thermoelectric, and electrokinetic cross effects.