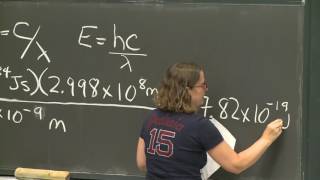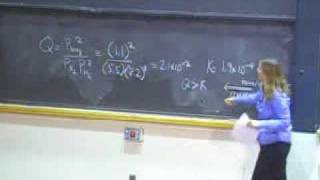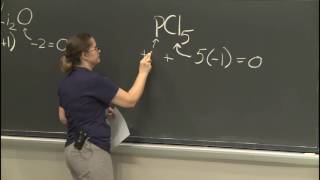UeduGPTs
Jupyters
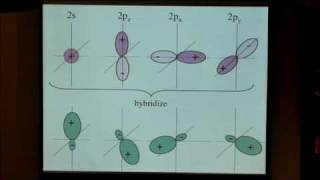
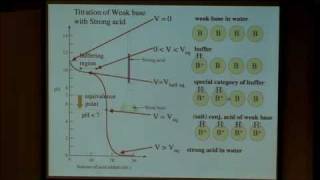
This course provides an introduction to the chemistry of biological, inorganic, and organic molecules. The emphasis is on basic principles of atomic and molecular electronic structure, thermodynamics, acid-base and redox equilibria, chemical kinetics, and catalysis.
In an effort to illuminate connections between chemistry and biology, a list of the biology-, medicine-, and MIT research-related examples used in 5.111 is provided in Biology-Related Examples.
AcknowledgementsDevelopment and implementation of the biology-related materials in this course were funded through an HHMI Professors grant to Prof. Catherine L. Drennan. Videos and captioning were made possible and supported by the MIT Class of 2009.
| 來源 | MIT 開放式課程 |
| 科系 | Chemistry |
| 語言 | English |
| 影片數 | 107 |